Leave Your Message
In the evolving landscape of healthcare, quality Medical Equipment plays a crucial role. Experts emphasize the importance of reliability and innovation in this sector. Dr. Emily Chen, a renowned expert in medical technologies, states, "The future of healthcare relies on accessible, high-quality Medical Equipment." This highlights the growing demand from global buyers seeking superior products.
China has emerged as a significant player in the Medical Equipment industry. Its advancements and production capabilities attract attention from various international markets. However, challenges remain. Buyers often face difficulties in identifying reliable suppliers amid a sea of options.
Moreover, understanding regulatory standards and product certifications is essential. Without proper knowledge, buyers may encounter risks. Hence, thorough research and expert guidance are critical. The journey to sourcing quality Medical Equipment is complex but rewarding.

China is becoming a powerhouse in the medical equipment sector, attracting global buyers looking for high-quality products. The country's largest manufacturers are investing heavily in innovation to stay competitive. According to a recent report, China accounted for 27% of the global medical device market in 2022. This figure is expected to increase as technology advancements continue.
Manufacturers in China are notably focusing on areas like imaging equipment, surgical devices, and telemedicine products. Some innovative solutions include AI-driven diagnostic tools and portable monitoring devices. However, not all products meet international standards. Reports show that about 10% of Chinese medical devices face challenges regarding quality assurance. This reality underscores the need for buyers to conduct thorough due diligence when selecting suppliers.
Furthermore, collaboration with research institutions is becoming common. Such partnerships aim to improve product quality and ensure regulatory compliance. While the growth of the medical device industry in China is promising, it raises questions about sustainability and ethical production practices. Buyers should remain vigilant, prioritizing transparency in the supply chain and seeking reliable partnerships.
China has become a pivotal player in the global medical equipment market. The country is known for a variety of devices, from imaging machines to surgical instruments. However, the quality of these products is often questioned. Understanding the quality standards in Chinese medical equipment production is crucial for buyers.
In recent years, China has taken substantial steps to align its manufacturing processes with international standards. The government has implemented stricter regulations and quality control mechanisms. This has led to improved reliability in products. Still, challenges remain. Some manufacturers overlook detailed compliance. This can lead to variations in quality. Buyers must be vigilant and assess each supplier's adherence to standards.
Many Chinese manufacturers are now investing in research and development. They are working to enhance their product lines and ensure safety. Yet, this progress is not uniform. Discrepancies can exist between regions and companies. It is vital for international buyers to engage in thorough research. Establishing partnerships with reliable suppliers can help mitigate risks. Quality assurance must be a priority for anyone sourcing medical equipment from China.
| Product Category | Quality Standard | Certification Body | Common Applications | Export Markets |
|---|---|---|---|---|
| Diagnostic Equipment | ISO 13485 | TÜV SÜD | Hospitals, Clinics | North America, Europe |
| Surgical Instruments | CE Mark | SGS | Operating Rooms | Australia, Asia |
| Patient Monitoring Systems | ISO 15197 | Intertek | ICU, Emergency Departments | Africa, Middle East |
| Therapeutic Equipment | MDD 93/42/EEC | Bureau Veritas | Rehabilitation Centers | Latin America, Europe |
| Laboratory Equipment | ISO/IEC 17025 | CNAS | Research Labs | Asia, North America |
China has established itself as a key player in the medical equipment industry. Various categories of medical devices sourced from China are gaining traction among global buyers. One notable category is diagnostic imaging equipment. Hospitals are increasingly relying on advanced ultrasound and X-ray machines to improve patient outcomes. These devices are often designed with the latest technology and competitive pricing.
Another critical category is surgical instruments. High-quality tools, including scalpels and forceps, are essential in operating rooms worldwide. Many manufacturers in China adhere to international standards, ensuring reliability and safety. However, buyers must conduct thorough research to verify the credibility of suppliers. Some factories may not meet strict quality controls.
Personal protective equipment (PPE) remains a prominent category. Demand surged during the pandemic, showcasing the importance of this sector. Buyers should remain vigilant regarding the sourcing process. There have been instances of subpar quality entering the market. Ensuring proper certifications and audits is essential for reliable procurement.
Evaluating the quality of medical equipment from China involves several critical factors. As global demand for medical devices rises, understanding the nuances of quality assessment is essential. The market reached a value of approximately $22 billion in 2022, with imports and exports growing exponentially. This surge emphasizes the need for buyers to be diligent.
One key aspect is certification. Look for internationally recognized certifications such as ISO 13485. This standard demonstrates a commitment to quality management systems. In addition, checking for compliance with CE marking ensures the equipment meets European safety standards. These markers are vital for assessing credibility. However, not all manufacturers may meet these benchmarks, prompting the need for thorough research.
Furthermore, testing and validation processes should not be overlooked. Reports indicate that 30% of imported medical devices have inconsistencies in performance. Engaging third-party testing labs can provide further verification of equipment reliability. Additionally, considering user feedback and reviews can offer real-world insights into product performance. This method allows buyers to identify potential issues and reflect on best practices in their equipment selection process.
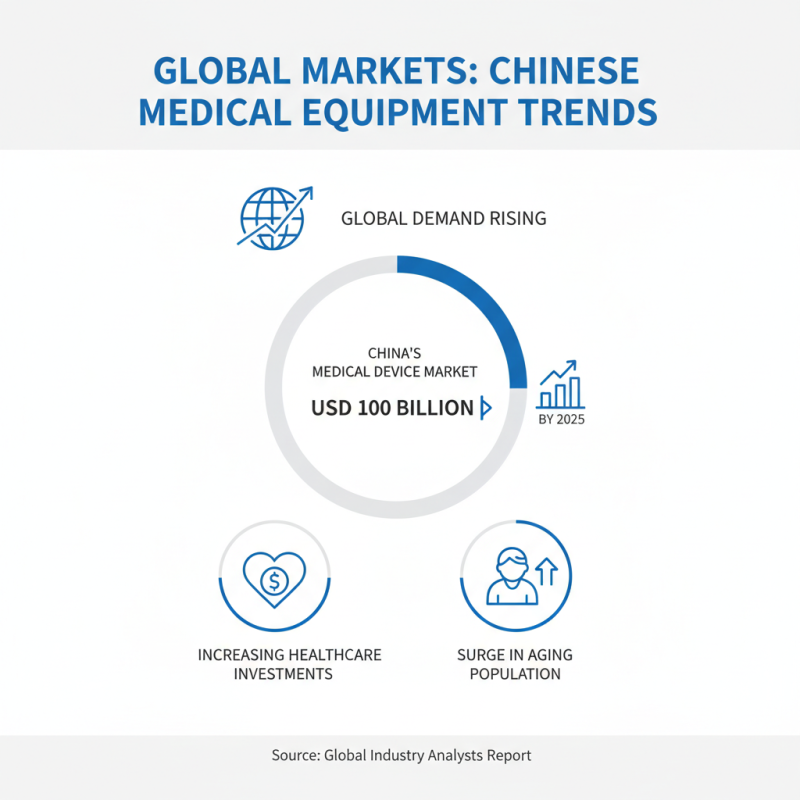
The global demand for Chinese medical equipment is rising steadily. According to a recent report by the Global Industry Analysts, the medical device market in China is projected to reach USD 100 billion by 2025. This growth is driven by increasing healthcare investments and a surge in the aging population.
Emerging trends indicate a shift towards advanced medical technologies. Telemedicine and wearable devices are becoming more common. A McKinsey report suggests that 70% of global healthcare consumers are willing to adopt telehealth solutions. However, there is still a gap in regulatory frameworks for quality assurance. This raises concerns about product reliability in foreign markets.
Data reveals that quality control remains a persistent challenge in manufacturing. A significant portion of equipment is criticized for lacking international standards. Buyers must be vigilant and conduct thorough due diligence. Increased collaboration between global entities and local manufacturers is essential. It fosters innovation but also highlights the need for transparency and diligence in quality assessments.